Composition Of Liquid And Vapour Phases. Theoretical. Part 2
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Composition Of Liquid And Vapour Phases. Theoretical. Part 2
Infinitely Miscible Liquids
In the case of liquids which are miscible in all proportions, it is only, as has already been pointed out, when no appreciable change of volume or temperature occurs on admixture that the vapour pressures and the boiling points can be accurately calculated. The relation between the composition of the mixed liquid and that of the vapour evolved from it appears also to be a simple one only when the vapour pressure of 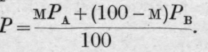 the mixture is accurately expressed by the formula
the mixture is accurately expressed by the formula
Formula Of Wanklyn And Berthelot
Many attempts have been made to find a general formula to represent the relation between the composition of liquid and of vapour. In 1863, Wanklyn 1 and Berthelot2 arrived independently at the conclusion that the composition of the vapour depends (a) on that of the liquid, (b) on the vapour pressures of the pure components at the boiling point of the mixture, (c) on the vapour densities of the components.
1 Konowaloff, "On the Vapour Pressures of Mixtures of Liquids," Wied. Ann., 1881,14, 34.
Calling m'A and m'B the relative masses of the two substances in the vapour, mA and mB their relative masses in the liquid, DA and DB their vapour densities and PA and PB their vapour pressures, the formula would be

In 1879, Thorpe 3 observed that carbon tetrachloride and methyl alcohol form a mixture of minimum boiling point, and that, for this particular mixture, when equal to unity.

is approximately
Brown's Formula
The subject was investigated experimentally by F. D. Brown in 1879-1881,4 and he found that the Wanklyn-Berthelot formula could certainly not be accepted as generally true. A better result was obtained with the formula but the agreement between the calculated and observed results was still closer when a constant, c, was substituted for the ratio PA/PB.
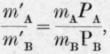
Applicability Of Formula
Brown's formula, is not generally applicable to liquids which are miscible in all proportions, and it is obvious that it cannot be true for two liquids which form a mixture of constant boiling point. The experimental evidence, however, which has so far been obtained, points to the conclusion that Brown's law is true for those infinitely miscible liquids for which the relation holds good.

The number of such cases so far investigated is, perhaps, too small to allow of the statement being definitely made that the law is true in such cases, but the evidence is certainly strong, for not only does
1 Wanklyn, " On the Distillation of Mixtures : a Contribution to the Theory of Fractional Distillation," Proc. Roy. Soc., 1863, 12, 534.
2 Berthelot, " On the Distillation of Liquid Mixtures," Compt. rend., 1863, 57, 430.
3 Thorpe, "A Contribution to the Theory of Fractional Distillation," Trans. Chem. Soc, 1879, 35, 544.
4 Brown, " The Comparative Value of Different Methods of Fractional Distillation," Trans. Chem. Soc, 1879, loc tit. ; 1880, 37, 49 ; " On the Distillation of Mixtures of Carbon Disulphide and Carbon Tetrachloride," 1881, 39, 304; "Fractional Distillation with a Still-head of Uniform Temperature," 1881, 39, 517.
Brown's formula hold good, within the limits of experimental error, for the two pairs of liquids investigated for which the relation between vapour pressure and molecular composition is represented by a straight line, but also, in other cases, it is found that the closer the approxima-tion of the pressure-molecular composition curve to straightness, the smaller is the variation in the value of the "constant " c.
Zawidski's Results
Strong evidence is afforded by two pairs of liquids, ethylene and propylene dibromides and benzene and ethylene dichloride, investigated by Zawidski.1
In the following tables are given the observed vapour pressures and those calculated from the formula also the observed molecular percentages of A in the vapour and those calculated from the formula


This formula may, of course, be written where m'a, m'b, ma and mb represent gram-molecules instead of grams.

Table 23
A = Propylene dibromide : B = ethylene dibromide ; c = 1*31 ; Temperature = 85.05° ; PB/PA at 85° = 1.357.
Molecular percentage of A in liquid. | Vapour pressures. | Molecular percentage of A in vapour. | ||||
Observed. | Calculated. | A. | Observed. | Calculated. | A. | |
0.00 | 172.6 | 172.6 | 0.0 | ... | ... | |
2.02 | 171.0 | 171.7 | + 0.7 | 1.85 | 1.55 | -0.30 |
7.18 | 168.8 | 169.3 | + 0.5 | 6.06 | 5.60 | -0.46 |
1475 | 165.0 | 165.9 | + 0.9 | 12.09 | 11.66 | -0.43 |
22.21 | 161.6 | 162.5 | + 0.9 | 18.22 | 17.89 | -0.33 |
29.16 | 158.7 | 159.4 | + 0.7 | 23.50 | 23.90 | + 0.40 |
30.48 | 158.9 | 158.8 | -0.1 | 23.96 | 25.08 | + 1.12 |
40.62 | 154.6 | 154.2 | -0.4 | 34.25 | 34.31 | + 0.06 |
41.80 | 153.4 | 153.6 | + 0.2 | 34.51 | 35.41 | + 0.90 |
52.63 | 149.6 | 148.7 | -0.9 | 45.28 | 45.89 | + 0.61 |
62.03 | 143.3 | 144.4 | + 1.1 | 55.35 | 55.50 | + 0.15 |
72.03 | 140.5 | 139.9 | -0.6 | 65.86 | 66.28 | + 0.42 |
80.05 | 136.8 | 136.3 | -0.5 | 74.94 | 75.39 | + 0.45 |
85.96 | 133.9 | 133.6 | -0.3 | 82.45 | 82.38 | -0.07 |
91.48 | 130.9 | 131.1 | + 0.2 | 89.50 | 89.13 | -0.37 |
93.46 | 130.2 | 130.2 | 0.0 | 92.31 | 91.60 | -0.71 |
96.41 | 128.4 | 128.8 | + 0.4 | 96.41 | 95.35 | -1.06 |
98.24 | 127.3 | 128.0 | + 0.7 | 99.39 | 97.71 | -1.68 |
100.00 | 127.2 | 127.2 | 0.0 | ... | ... | ... |
The agreement between the observed and calculated percentages of propylene dibromide in the vapour is not altogether satisfactory,
1 Zawidski, " On the Vapour Pressures of Binary Mixtures of Liquids,"Zeitschr. physik. Chem., 1900, 35, 129.
G but Zawidski states that the errors of experiment were much greater for this pair of substances than for others, owing to the small quantity of material at his disposal. The last two observed molecular percentages of propylene dibromide in the vapour, 9641, and 99.39, are obviously too high; the last should indeed be less than 98.24.
It will be seen later that when there is a real variation in the values of c, it is in the nature of a steady rise or fall from A=0 to A = 100 per cent, whereas, in this case, the calculated values of c would be low at each end of the table.
The value of c, 1.31, does not differ greatly from the ratio of the vapour pressures, 1.357.
This series of experiments is of special interest because the two liquids are very closely related. Zawidski, however, found that the relation held good with considerable accuracy for mixtures of benzene and ethylene dichloride, and the data for this pair of liquids are therefore given in full.
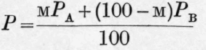
Continue to:


