General Remarks. Part 2
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
General Remarks. Part 2
Second Case
Figure 76 represents the separation of a liquid of constant boiling point from a mixture which contains only much more volatile impurities. The separation is an easier one than the last since it is the least volatile component that is to be isolated. If the temperature remains constant for a considerable time and shows no rise whatever at the end of the distillation, the last portion need not be redistilled, and in subsequent fractionations the distillation may be stopped as soon as the maximum temperature has been reached, and the residue may be taken as pure.
Here, again, it is possible that we may be dealing with a mixture of maximum boiling point as, for example, in the separation of the binary mixture of chloroform and methyl acetate from a slight excess of the ester, but it is less probable, for mixtures of maximum boiling point are not met with so frequently as those of minimum boiling point. It is also possible, though less likely, that we might have a mixture of minimum boiling point containing neither substance in excess but contaminated with a more volatile impurity.
Third Case
A curve like that in Fig. 77, would result from the distillation of a liquid containing an impurity, the boiling point of which was not much higher than that of the chief component.

Weight of Distillate Fig. 76.
The separation in such a case would be much more difficult, and several fractionations would be required before the boiling point of the first fraction became quite constant. When the boiling points of the components of a mixture are very near together and the chemical relationship is not very close, mixtures of constant boiling point are not unlikely to be formed; that is the case, for example, with carbon tetrachloride and benzene, and it would be practically impossible to separate either pure tetrachloride or the mixture of constant boiling point from any mixture of the two substances, although benzene, if present in large excess in the original mixture, could be separated in a pure state by repeated fractionation.
Fourth Case
A curve such as that in Fig. 78, would represent the behaviour on distillation of any mixture -such as that last-named with benzene in large excess - in which the chief component was the less volatile, but in which the difference between the boiling points of the components was small. The separation of the chief component would almost invariably be easier than in cases represented by Fig. 77. Indeed, Fig. 78 bears the same relationship to Fig. 77 as Fig. 76 to Fig. 75.

Fig. 77.
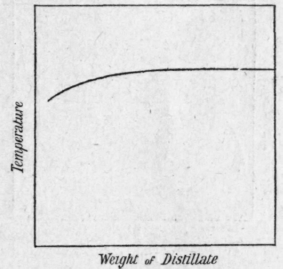
Fig. 78.
Here, again, the highest fraction might consist of a mixture of maximum boiling point such as chloroform-methyl acetate ; (b.p. 64.5°) with chloroform (b.p. 60.5°) in excess ; or, less probably, of a mixture of minimum boiling point containing a more volatile impurity, as, for example, the isopropyl alcohol-water mixture (b.p. 80.35°) with a little ethyl alcohol (b.p. 77.3°).
Fifth And Sixth Cases
The curves in Figs. 79 and 80 represent the distillation of liquids which contain impurities of both greater and less volatility. In the case of Fig. 79, the boiling points of these impurities are far removed from that of the substance to be separated ; in the case of Fig. 80 they are near it. Such separations as these have very frequently to be carried out.
When, for example, benzene is nitrated by treatment with nitric and sulphuric acids, some of the benzene is usually unacted on, while a certain amount of dinitrobenzene is formed. Here the boiling point of the chief component, nitrobenzene, is very much higher than that of benzene and far below that of dinitrobenzene, and the separation is therefore an easy one. If pure benzene is used for the preparation, and the mixture is distilled through a "pear" still-head with, say, 12 bulbs, the collection of pure nitrobenzene may be commenced after the second or third fractionation.
When, however, the boiling points are not so far apart, as, for example, in the separation of ethyl acetate from methyl and propyl acetate, the process is tedious if an ordinary apparatus is used (p. 105), and even with a 5-column "evaporator" still-head several fractionations are required (p. 159), for the middle substance is always more difficult to separate than the others.
If the boiling points of the components are very near together (Fig. 80) the separation of the middle substance is extremely troublesome, and there is greater probability that mixtures of constant boiling point may be formed than in the cases previously considered.

FlG. 79.

Fig. 80.
Other Cases
Other cases than those referred to may be met with; for example a liquid may contain only impurities of greater volatility, but the boiling points of some of these may be near, those of others far below that of the chief component. The most volatile impurities in such a case will be easy to remove, while the less volatile will only be eliminated with difficulty and the distillation will correspond to those represented by the curve in Fig. 78 rather than in Fig. 76.
2. The Separation of the Components from a Mixture of Known Qualitative Composition
Two Components
The chief points to be considered are (a) the boiling points of the pure components, and the difference between them ; (6) the chemical relationship of the components ; (c) the form of the boiling point-molecular composition curve, if it can be ascertained.
Continue to:


