Chapter XIX. General Remarks
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter XIX. General Remarks
Purposes for which Fractional Distillation is required. Interpretation of Experimental Results. Choice of Still-head, Number of Fractions, etc.
Purposes for which Fractional Distillation is required.
1 Fractional distillation may be employed for various purposes, of which the following are among the most important:1. The isolation of a single substance from a mixture with the smallest possible loss.
2. The separation of the components from a mixture of known qualitative composition.
3. The determination of the quantitative composition of a mixture of which the qualitative composition is already known, or the determination of the quantitative composition of a mixture of constant boiling point.
4. A general study of the qualitative and quantitative composition of a complex mixture when only the general nature of the chief components is known, or when it is definitely known that certain substances are present but it is not known what others there may be.
1. The Separation of a Single Substance from a Mixture with the Smallest Possible Loss
When a pure substance is to be isolated from a mixture, the procedure will be somewhat simplified if the true boiling point of the chief component is already known.
In any case a careful record should be made of the temperature range and weight of each fraction, and we may then either calculate the or plot the total weights of distillate against the final temperatures of the tractions.
Much time will be saved if an improved still-head is employed ; for liquids of high boiling point, the "pear," and for volatile liquids the "evaporator" apparatus may be especially recommended.
Interpretation of Results. First Case. - If the results obtained by the distillation of the liquid under examination give a curve similar in form to Fig. 75, we may conclude that there is present a relatively small quantity of some impurity of much higher boiling point, but that impurities more volatile than the chief component are absent.
As the temperature shows no appreciable rise until towards the end of the distillation, the first fraction will have a perfectly constant boiling point when redistilled, and we may conclude that it will most probably consist of the pure substance required. In the second and subsequent fractionations the greater part of the distillate from the first fraction may, in each case, be taken as most probably pure and will not require to be redistilled. The separation of the liquid of constant boiling point will in such a case as this be an easy one.
Proofs Of Purity
Even, however, if the true boiling point of the pure substance is known, and if the observed boiling point agrees with it, we cannot be absolutely sure of the purity of the liquid unless we know - from the method of preparation - that there is no possibility of the formation of a mixture which distils without change of composition at practically the same temperature as the pure liquid. If we can be sure that the original mixture contains only substances which are very closely related to each other, such as members of a homologous series, the problem will be much simplified, for it may then be concluded with certainty that no mixture of constant boiling point can be formed.
If any doubt exists and if the specific gravity of the pure liquid is accurately known, it is best to determine that of the distillate : if both the boiling point and the specific gravity of the distillate agree with those of the pure substance, we may conclude with confidence that the distillate is really pure. Instead of the specific gravity, the refractive index, the vapour density, the melting point (if the liquid can be easily solidified), or some other physical constant may be determined for comparison, or a chemical analysis of the liquid may be made.
Hexane And Benzene
Suppose, for example, that the original liquid consisted chiefly of normal hexane, and that we were unacquainted with the previous history of the specimen ; there would be the possibility of the presence of some benzene. A mixture of normal hexane and benzene containing as much as 10 per cent of the aromatic hydrocarbon boils at almost exactly the same temperature as pure hexane, and therefore the presence of the benzene could not be detected, and the impurity could not be removed, by fractional distillation. The high specific gravity of the distillate would, however, show that some other substance besides normal hexane was present.
If, on the other hand, it were known that the hexane had been prepared synthetically from pure propyl iodide by the action of sodium, there would be no possibility of the presence of benzene.
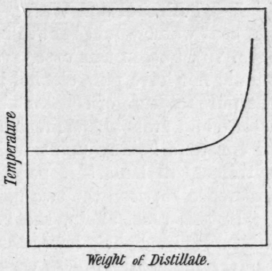
Fig. 75.
Ethyl Alcohol And Water
Ethyl alcohol containing, say, 15 or 20 per cent water would behave in the manner indicated by the same curve (Fig. 75), but, in this case, even with the most efficient still-head, we should not have pure alcohol in the first part of the distillate, nor even the pure mixture of constant boiling point, but a mixture containing at least 5 and probably as much as 7 or 8 per cent of water, for the mixture of constant boiling point which contains 95.6 per cent of alcohol is extremely difficult to separate from water, although there is a wide difference between the two boiling points.
In this case, again, the boiling point of the binary alcohol-water mixture is so slightly lower than that of pure alcohol that the reading of the thermometer - unless very accurate - would hardly be sufficient to distinguish with certainty between the two.
Isopropyl Alcohol And Water
With isopropyl alcohol and water, similar results would be obtained except that the difference between the boiling points of the pure alcohol and of the binary mixture is sufficiently great for the two to be distinguished without difficulty by the observed temperature, and also that the binary mixture can be separated in a pure state from water.
Continue to:


