Chapter X. Modifications Of The Still-Head
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter X. Modifications Of The Still-Head
Object Of Modifications
It is evident from what has been stated in Chapter VII (Directions For Carrying Out A Fractional Distillation). that the process of fractional distillation with an ordinary still-head is frequently an exceedingly tedious one, and many attempts have been made to modify the still-head in such a manner as to bring about a more complete separation in a single operation.
The idea of improving the still-head is by no means a new one, for in a book entitled Philosophorum, sen liber de Secretis Naturce, by Philip Ulstadius, 1553, there is an illustration of a five-headed still (Fig. 41). When dilute spirit was distilled through this "alembic " it is clear that the alcohol collected in the uppermost receiver must have been considerably stronger than that in the lowest.
Great progress was made in the improvement of the still-head in commerce - notably for the better separation of alcohol from weak spirit - before any advance was made in the laboratory, and, indeed, the improved still-heads first employed in the laboratory were, for the most part, merely adaptations of those already in use on the large scale.
It may, however, be convenient to consider first those forms of apparatus which are suitable for laboratory purposes.
The object of the modifications which have been made in the still-head is to bring about a more complete separation of the components of the mixture in a single distillation, a process of fractionation - more or less effective according to the form of apparatus and the rate of distillation - taking place in the still-head.1

Fig. 41. - Five-headed still described by Ulstadius, 1553.
1 Young, " On the Relative Efficiency and Usefulness of Various Forms of Still-head for Fractional Distillation, with a Description of some New Forms possessing Special Advantages," Trans. Chem. Soc, 1899, 75, 679.
Inefficiency Of Plain Vertical Still-Head
Of all possible forms, the plain vertical still-head is the least efficient. During the distillation, as the vapour rises up the cylindrical tube, the outer parts of it come in contact with the condensed liquid flowing down the sides of the tube. Since the tube is constantly losing heat by radiation and by conduction to the surrounding air, this liquid is slightly cooled and fresh condensation constantly goes on, the outermost layer of vapour probably condensing almost completely without much change of composition. The central portion of vapour rises rapidly up the tube and can only reach the liquid by diffusion or by convection currents, and much of it may pass through the still-head without reaching the liquid at all; the condensed liquid, on the other hand, flows rapidly down the vertical walls of the tube, back to the still. In order, however, that a satisfactory separation of the components of the mixture may take place, as much of the vapour as possible should be brought into intimate contact with the condensed liquid, so that a state of equilibrium, as regards composition, may be brought about.
Brown's Formula
Let us suppose, to facilitate the consideration of what occurs, that we are dealing with a mixture of two closely related compounds, to which Brown's formula is applicable, and that m' and m refer to the weights of vapour and liquid respectively. Also let us suppose that, in the first place, equal weights of the two substances are taken, so that mA = mB.
Then the composition of the first small quantity of vapour formed will be given by the equation and the percentage of b in the vapour will be

If a distillation bulb with exceedingly short still-head be employed, this will practically be the percentage of b in the first portion of distillate.
Theoretically Perfect Still-Head
Suppose, now, that in the lower part of a long and theoretically perfect still-head condensation goes on, and that the condensed liquid remains in this part of the tube until its weight is, say, 100 times as great as that of the residual un-condensed vapour in the same part of the tube, the total weight of liquid and vapour being, say, 1/1000 of that of the mixture taken. We may then assume
1. That the composition of the liquid and vapour (taken together) in the still-head will be practically the same as that of the vapour first formed, and
2. That the composition of the condensed liquid will not differ sensibly from this, or it the residual vapour is brougnt into thorough contact with the condensed liquid, its composition will practically be given by the equation and the percentage of B in it will be

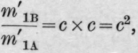
If we suppose, as a theoretical case, that the still-head is divided into n sections, and that the distillation goes on in such a manner that perfect equilibrium between vapour and condensed liquid is established in each section, and, lastly, that the condensed liquid in any section has the same composition as the vapour in the section below, then we should have, for the composition of the vapour in the nth. section, when the distillate begins to come over,
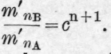
If we take c = 2.5, as in the case of benzene and toluene, and start with equal weights of the components, the composition of the vapour in the still and in the first nine sections would be as follows: Table 47
No. of Section. | Percentage of B. | No. of Section. | Percentage of B. |
Still | 71.43 | 5 | 99.59 |
1 | 86.21 | 6 | 99.84 |
2 | 94.55 | 7 | 99.93 |
3 | 97.51 | 8 | 99.97 |
4 | 98.99 | 9 | 99.99 |
Such an arrangement is not realisable in practice, but the attempt should be made to approach as closely to it as posssible.
Continue to:


