Chapter XIII. Continuous Distillation
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter XIII. Continuous Distillation
Continuous distillation of wort has long been carried out on the large scale by means of the Coffey still (p. 167) and others of the same type, but it is only comparatively recently that any attempt has been made to devise a process of the kind suitable for laboratory purposes.
Carveth's Apparatus
Carveth1 suggests that by maintaining two parts of a system at different temperatures, corresponding to the boiling points of the two components of a mixture, it should be possible to effect continuous separation, especially if use were made of dephlegmating intercepts. He describes an apparatus used by Derby, in 1900, for mixtures of alcohol and water. It consists (Fig. 56) of a long block-tin tube surrounded at its lower end, a, by the vapour of boiling water, at its upper end, b, by vapour from boiling alcohol, and filled with intercepts. The mixture of alcohol and water to be distilled was slowly dropped in at c, the vapour passing to the condenser at D, and the residue through the trap at e. Carveth, however, gives no details regarding the length or diameter of the tubes or the nature of the intercepts.
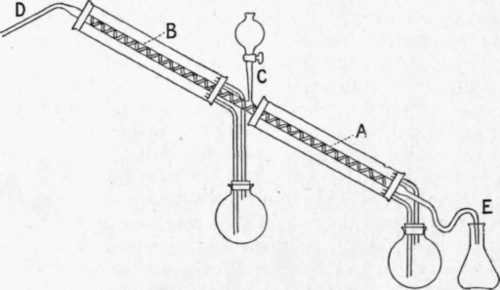
Fig. 56. - Carveth's still for continuous distillation.
So far as the lower half of the still-head is concerned, there is no objection to the use of steam as a jacket, the object aimed at being to keep the temperature up to the boiling point of water in order that the alcohol may be vaporised as completely as possible ; the great amount of heat evolved by the condensation of steam makes that substance a very efficient heating agent. But the use of the vapour of alcohol for the upper part of the still-head is wrong in principle, and it is a mistake to suppose that this part would thus be maintained "at a temperature corresponding to the boiling point of alcohol." What is here required is to keep the temperature as far as possible from rising above the boiling point of alcohol, in order that the vapour of water may be condensed as completely as possible, while that of the alcohol passes on to the condenser. This can be done by surrounding the still-head with a liquid which is kept at the required temperature by suitable means, but if a vapour, such as that of the more volatile component, is used as a jacket, it easily becomes superheated and the still-head is not prevented from rising in temperature at all; indeed the effect is very similar to that produced by covering the tube with cotton-wool - condensation is somewhat diminished and so also is the efficiency.
1 Carveth, "Studies in Vapour Composition," Part II. Journ. Phys. Chem., 1902, 6, 253.
Carveth makes the general statement that the percentage of alcohol by weight in the residue was found to average about 0.5 when working carefully, but in five cases for which he gives details the percentage varied from 1.5 to 2.0. These five distillations were carried out with extreme slowness, the average rate being only 7.5 grams of distillate per hour.
Again, the percentage of alcohol in the distillate is stated to have varied from 90.6 to 93.9 in four cases, but in the five very slow distillations referred to above the percentages varied from 76.7 to 89.9. It does not appear possible to form any very definite idea of the efficiency of the apparatus from the data given.
Lord Rayleigh's Apparatus
Lord Rayleigh1 describes an apparatus similar in principle to Carveth's except that the temperatures of both parts of the still-head are regulated by liquids and not by vapours.
The apparatus consists of a long length (12 metres) of copper tubing, 15 mm. in diameter, arranged in two spirals which are mounted in separate pails. For the distillation of mixtures of alcohol and water the lower and longer spiral was heated by boiling water, the upper one by water maintained at a suitable temperature, usually 77°. The spirals were connected by a straight glass or brass tube of somewhat greater bore, provided with a lateral junction through which the mixture could be introduced. With the exception of the two extremities, the whole length of tubing sloped gently and uniformly upwards from near the bottom of the lower pail to the top of the upper pail, where it turned downwards and was connected with an ordinary Liebig's condenser. The lower end of the tubing was, if necessary, connected with an air-tight receiver heated to 100°.
1 Rayleigh, " On the Distillation of Binary Mixtures," Phil. Mag., 1902, [VI.], 4, 521.
The mixture was introduced at such a rate that it fell in a rapid but visible succession of drops and, when rich in alcohol, it was previously heated. Mixtures containing 20, 40, 60 and 75 per cent of alcohol were distilled, and in all cases the water, collected in the lower receiver, was nearly pure, never containing more than 0.5 per cent of alcohol. The distillate varied but little in strength and contained from 89 to 90.3 per cent of alcohol.
It seems probable that such a continuous process may prove very useful for mixtures which separate into only two components and when the quantity to be distilled is large.
Continuous Separation Of Three Components
It would, moreover, be possible to devise an arrangement by which three components could be separated in a similar manner, but it would be necessary to have two coils for the middle substance ; the form of apparatus required is shown diagrammatically in Fig. 57. Suppose, for example, that the liquids to be separated were methyl, ethyl and propyl acetates (b.p. 574°, 77.15° and 101.55° respectively). The highest bath would be kept at 57.1°, the middle one at 77.15° and the lowest at 101.55°. The mixture would be introduced slowly through the funnel at a, into the still B, where it would be boiled, and the mixed vapour would enter the still-head at c. The propyl acetate would be collected in the receiver D, which might be heated by a ring burner. The vapour in passing upwards through the first coil in the middle bath would be freed, more or less completely, from propyl acetate and the mixture of ethyl and methyl acetates would enter the top of the second coil, chiefly in the form of vapour. The condensed liquid which reached the bottom of the coil, and was collected in e, would be nearly pure ethyl acetate; while the vapour that reached the top of the coil in the highest bath, and was condensed in f, would be nearly pure methyl acetate.
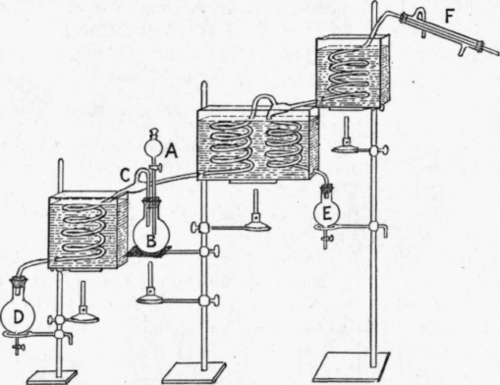
Fig. 57. - Still for continuous distillation of three components. (Adapted from Lord Rayleigh's still.).
It should thus be theoretically possible to separate all three liquids in a pure state, and the components of a still more complex mixture should be separable, if there were as many baths as components kept at the boiling points of those components, and two coils in each bath except the lowest and highest.
Messrs. E. Barbet et Fils et Cie, of Paris, supply an efficient laboratory apparatus which can be used for either continuous or discontinuous distillation.
Continue to:


