Gas. Part 3
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Gas. Part 3
Its composition, according to Profs. Silliman and Wurtz, is: marsh gas, 82.41; carbonic acid, l0.11; nitrogen, 4*31; oxygen, 0.23; illuminating hydrocarbon, 2.94. It was carried in a wooden main to the city of Rochester, a distance of 24 miles, in 1870, for the purpose of illuminating the city, but the experiment was a failure. Such gas has also been known to issue from beneath peat bogs, as that of Chatmoss, near the Manchester and Liverpool railway in England. These natural gases are of variable composition, light car-buretted hydrogen or marsh gas being usually the principal constituent.- Coal Gas. The first artificial production of illuminating gas from coal is due to Dr. Hales or the Rev. John Clayton, rector of Crofton, Wakefield, Yorkshire, England. Dr. Hales describes in a book published in 1727, called Vegetable Statics,11 an experiment by which he obtained 180 cubic inches of inflammable air from 158 grains of Newcastle coal. In 1659 Mr. Thomas Shirley communicated a paper to the royal society on an inflammable gas which issued from a well near Wigan in Lancashire; and the Rev. John Clayton nearly a century later, in examining the locality, found that the gas issued from a bed of coal.
The experiment of subjecting the coal to heat was made, with the result of obtaining an illuminating gas, which, being collected in bladders, could be burned in jets. An account of these experiments was communicated to the royal society in 1739. Dr. Rickel, professor of chemistry at Wurzburg, in 1786 lighted his laboratory with gas made by the dry distillation of bones; and in the same year Earl Dundonald of Scotland, in obtaining tar from the distillation of coals, also collected an illuminating gas, with which he lighted Culross abbey by way of experiment. The first practical attempt at gas lighting is however generally accredited to William Murdoch, who in 1792 used coal gas for lighting his workshops at Redruth in Cornwall. His invention remained unknown till about 1802, when it was introduced at the Soho foundery of Boulton and Watt, near Birmingham. In the mean time Le lion, a Frenchman, used gas made from wood for lighting his house, and was therefore considered by the French as the inventor of gas lighting. In 1804 Mr. Murdoch lighted the mills of Phillips and Lee at Manchester with gas which yielded an amount of light equal to that of 3,000 sperm candles.
In 1813 London bridge was lighted with gas, and about the same time it was introduced into the streets in that part of the city; but it was not introduced into Paris till 1820.-Bituminous coals, such as English cannel and boghead coals, Ohio cannel, and the caking coals of Pennsylvania, Maryland, and Virginia, are used in this manufacture. The following may be considered as the average composition of caking and cannel coals:
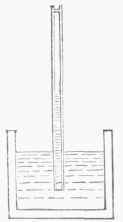
Fig.. 5.-Graham's Diffusiometer.
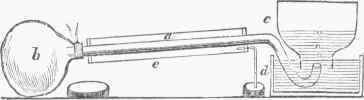
Fig. G.-Atiuolyser.
CONSTITUENTS. | Caking. | Cannel. |
Carbon........ | 87.752 | 75 25 |
Hydrogen........... | 5.239 | 5.50 |
Nitrogen........... | 1.702 | 1.61 |
Oxygen........... | 3.806 | 1383 |
Ash................ | 1.393 | 2.81 |
There are also small quantities of sulphur and iron, mostly in the form of iron pyrites. The nitrogen has its origin in the organic matter from which the coal is derived. When bituminous coal is heated to redness in the presence of air, it is principally converted into gases which unite with oxygen; but if air is excluded, as when the coal is confined in retorts, the gaseous products, unable to unite with oxygen, may be collected in receivers and burned in tubes. The products of the destructive distillation of bituminous coal consist of a great number of gases, liquids, and solids, which may be conveniently included under the following heads, according to an analysis by Bunsen:
Coke................. | 68.93 |
Tar................... | 12.23 |
Water | 7.40 |
Marsh gas............ | 7.04 |
Carbonic oxide..... | 1.13 |
Carbonic acid............ | 1.07 |
Oleflant gas......... | 0.78 |
Sulphuretted hydro. gen.................... | |
0.75 | |
Hydrogen........ | 0.50 |
Ammonia......... | 0.17 |
Nitrogen........... | 0.03 |
The olefiant gas here represents not pure heavy carburetted hydrogen or elayl, but a mixture, in variable quantities, of acetvlene, C2H2, elayl, C2H4, trityl, C3H6, ditetryl, C4H8, and several hydrocarbon vapors. There are also small quantities of cyanogen and sulpho-cyanogen. (See Distillation, Destructive.) -Upon the temperature to which the coal is subjected depend the products of distillation, which are formed by a rearrangement of the elements of the coal. The lower the heat, the less will be the weight of coke or carbonaceous residue in the retort, and therefore the greater will be the quantity of carbon which remains combined with hydrogen; but the hydrocarbons so formed will be mostly liquid and solid, and not gaseous. On the other hand, the higher the temperature the greater will be the weight of solid carbonaceous residue; and the proportion of permanent gases will be increased, and their levity also, in proportion to the heat, so that they may be almost entirely composed of hydrogen and carbonic oxide. A mean temperature has therefore to be employed by the gas manufacturer, and this will also depend upon the quality of the coal or other material used.
Practically it is impossible to subject the entire mass of coal to the most favorable conditions, because the different portions contained in a retort are necessarily subjected to different degrees of heat.-The manufacture of gas from coal, as well as from wood, petroleum, or resin, consists of three processes: 1, the distillation of the crude gas; 2, its separation from tarry and other condensible matters; 3, its purification from other deleterious and unpleasant gases. The distillation is performed in fire-clay retorts placed in furnaces, from five to ten retorts being placed in one furnace in large works, which may contain 100 furnaces. They are usually of a semi-cylindrical shape, somewhat like that of a sole drain tile, about 9 ft. in length, and from 16 to 20 in. in diameter. They were formerly made of cast iron, as fire clay was too porous and allowed the gas to escape; but since the introduction of apparatus for relieving the pressure, which will be described further on, fire-clay retorts are found sufficiently tight, and are much more durable. Moreover, they may be glazed and rendered more impervious than iron.
Continue to:


