Gas. Part 2
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Gas. Part 2
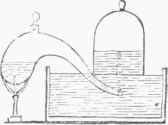
Fig. 1.-Collection by Displacement.
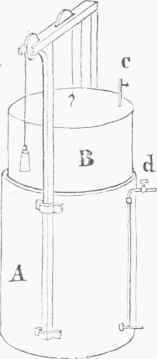
Fig. 2.-Gas-holder.
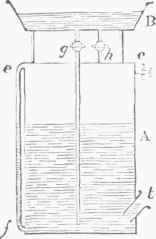
Fig. 3.-Gas-holder.
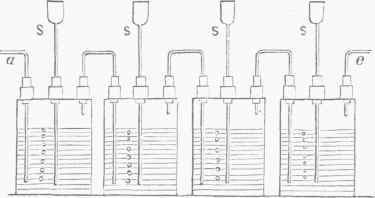
Fig. 4.-Woulfe's Bottles.
When the upper vessel contained hydrogen, the time occupied in diffusion was about 12 days; but when it contained air, oxygen, or nitrogen, several weeks were occupied in the complete diffusion. If a cylinder is filled with any gas and placed in a horizontal position, and an open tube bent at right angles is inserted at one end, turned upward if the gas is lighter than air, and downward if heavier, after a time the gas will escape from the cylinder and its place be occupied by air. The results given in the following table were obtained by Graham, and show the proportions of different gases which escaped from 100 volumes in four and in ten hours:
NAME OF GAS. | Sp. gr. | In 4h. | In 10 h |
Hydrogen......................................... | 1 | 81.6 | 94.5 |
Light carburetted hydrogen............ | 8 | 43.4 | 62.7 |
Ammonia.............................................. | 8.5 | 41.4 | 59.6 |
Olefiant gas............ | 14 | 34.9 | 483 |
Carbonic acid.......... | 22 | 316 | 47.0 |
Sulphurous acid............................ | 32 | 27.6 | 46.0 |
Chlorine............. | 35.4 | 23.7 | 895 |
It is here seen that the lighter gases are the more readily they escape, and that this tendency to diffusion is nearly in the inverse proportion of the square root of their densities. If the cylinder contains a mixture of gases, those which are the lightest and therefore the most readily diffusible will escape with the greatest rapidity. Vapors also diffuse themselves among one another and among the permanent gases in accordance with the same law. If the mouth of the tube, in the apparatus mentioned above, is closed with a porous substance like plaster of Paris or wood, and the cylinder is filled with hydrogen, this gas will escape much faster than the air will enter. It appears, therefore, that the smaller the calibre of the orifices through which the diffusion takes place, the greater will be the proportional rapidity of the transfusion of the lighter gas; a fact having a close relation to the phenomena of osmose, which it greatly aids in explaining. Graham has made extensive experiments on the diffusion of gases.
By using an instrument called a diffusion tube or diffusiometer, by means of which exact measurement could be made of the rate at which interchange took place, he found that diffusion through porous septa followed the same law as when the communication was by tubes of sensible diameter; that is, that the diffusibility of gases is in proportion to the square roots of their densities. The diffusiometer used by Graham (fig. 5) consists of a glass tube of about one inch calibre and one foot in length, and a vessel partly filled with mercury. One end of the tube is stopped with a plug of plaster of Paris, one fifth of an inch thick, which is formed by mixing the plaster into a paste with water, introducing it while in a plastic state, and allowing it to set and dry. When the tube is filled with hydrogen and its open end placed in the vessel of mercury, diffusion takes place through the porous plaster plug, the atmospheric air passing in and the hydrogen passing out; but the latter action is so much the more rapid that in three minutes the mercury will rise in the tube three inches above its level in the outer vessel, and in 20 minutes all the hydrogen will escape.
Graham afterward used compressed graphite, such as is used in making writing pencils, in place of the plaster of Paris, and considered it superior; but the results which he obtained with it did not alter the conclusions arrived at in the first series of experiments.-Atmolysis, or the Separation of Gases by Diffusion. When a mixture of gases passes through a graphite plate into a vacuum, a rapid separation takes place, in proportion to the difference of pressure. This method of separation may be performed with an apparatus called an atmolyser (fig. 6). A porous earthenware pipe, a, passes from a bag, b, containing the mixed gases, to the receiver c, over a pneumatic cistern. A large tube, e, surrounds the smaller one, and the space between them is kept as nearly vacuous as possible by means of an air pump exhausting through the tube d. A slight pressure upon the bag causes the gases to flow through the tube a, but the lighter ones will to a great extent pass through the sides of the tube and be removed by the air pumps. In an experiment made in this manner with atmospheric air, the proportion of oxygen was increased to 24.5 per cent. When a mixture of oxygen and hydrogen is used, the separation is much greater.
In one experiment an explosive mixture of one volume of oxygen with two of hydrogen, after atmolysis, contained only 9.3 per cent. of hydrogen, in which a taper burned without producing explosion. The diffusion of gases through one another was regarded by Dalton as a necessary consequence of the self-repulsive property of the particles of gaseous bodies, by reason of which each gas expands into the space occupied by the other as it would into a vacuum. But although the velocities with which they diffuse into each other are in proportion to those with which they rush into a vacuum through an aperture in a thin plate, still they do not strictly act as vacua to each other; for the diffusion of one gas into another is vastly slower than its passage into a vacuum.
III. Illuminating Gas. This is to a limited extent obtained from natural sources, but the great supplies are made from the following substances: 1, coal; 2, wood; 3, peat; 4, resin; 5, petroleum; 6, oils and fats; 7, water and coke. There are many localities where combustible gases have long been known to issue from the earth. Gas has been used in China for centuries, conveyed in bamboo tubes from fissures in salt mines in excavations from 1,200 to 1,600 ft. in depth. Near the Caspian sea in Asia there are several so-called eternal fires caused by gas issuing from the soil. In the Szalatna salt mine in Hungary illuminating gas constantly issues from a muddy clay contained between rock-salt strata. The village of Fredonia, N. Y., is lighted by gas which issues from bituminous limestone interspersed among the slates and sandstones of the Portage group. This gas consists of a mixture of marsh gas, CH4, and hydride of ethyl, C2H6. A flow of natural gas issued from a well which was sunk in 1865 at West Bloom-field, Ontario co., N. Y., for the purpose of obtaining petroleum.
Continue to:


