Boiling Points Of Mixed Liquids. Part 2
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Boiling Points Of Mixed Liquids. Part 2
Practical Application
Advantage is taken of the fact that the boiling point of a pair of non-miscible or slightly miscible liquids is lower than that of either pure component, to distil sub-stances which could not be heated to their own boiling points without decomposition, or which are mixed with solid impurities.
As a rule, water is the liquid with which the substance is distilled and the process is. commonly spoken of as "steam distillation." As an example, the commercial preparation of aniline may be described. Nitrobenzene is reduced by finely divided iron and water with a little hydrochloric acid, the products formed being ferrous chloride, magnetic oxide of iron and aniline. The aniline is distilled over with steam and the greater part of it separates from the distillate on standing. The aqueous layer, which contains a little aniline, is afterwards placed in the boiler which supplies steam to the still, so that in the next distillation the aniline is carried over into the still again.
The apparatus is shown diagrammatically in Fig. 21. A is the pipe by which steam is introduced into the still, and b is a rotating stirrer.1
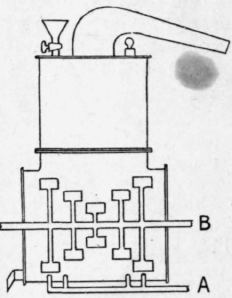
Fig. 21. - Aniline still.
Liquids Miscible In All Proportions
The boiling point of a mixture of two liquids which are miscible in all proportions can be calculated if the vapour pressure of the mixture agrees accurately with that given by the formula
The boiling points of such mixtures are, however, not so simply related as their vapour pressures to the molecular composition.
Calculation from Vapour Pressure and Composition.In order to calculate the boiling points of all mixtures of two such liquids under normal pressure, we should require to know the vapour pressures of each substance at temperatures between their boiling points. Thus chlorobenzene boils at 132.0° and bromobenzene at 156.1°, and we must be able to ascertain the vapour pressures of each substance between 132° and 156°.
The percentage molecular composition of mixtures which would exert a vapour pressure of 760 mm. must then be calculated at a series of temperatures between these limits, say every two degrees, by means of the formula where, in this case, P = 760.
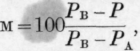
Lastly, the molecular percentages of A, so calculated, must be mapped against the temperatures, and the curve drawn through the points will give us the required relation between boiling point and molecular composition under normal pressure.
Closely Related Liquids
For such closely related substances as chlorobenzene and bromobenzene, the ratio of the boiling points on the absolute scale is a constant at all equal pressures, as is also the value of T . dp/dt, and it appears, so far as experimental evidence goes, that the ratio of the boiling point (abs. temp.) of any given mixture to that of one of the pure substances at the same pressure is also a constant at all pressures; it would therefore, strictly speaking, be sufficient to determine the vapour pressures of either substance through a wide range of temperature and the ratios of the boiling points (abs. temp.) of the other pure substance and of a series of mixtures to that of the standard substance at a single pressure, in order to be able to calculate the boiling point of any mixture at any pressure.
The vapour pressures of chlorobenzene and bromobenzene from 132° to 156° are given in the table below, also the values of PB-760, Pb --Pa and m.
For further information on the use of steam in distillation, reference may be made to Hardy and Richens, "Fractional Distillation by Means of Steam," Analyst, 1907, 32, 197 ; R. Zaloziecki, "Theory of Distillation by Steam," Petroleum Review, 3, 425; H. Golodetz, "Fractional Distillation with Steam," Zeitschr. phijsik. Chem., 1912, 78, 641.
Table 11
Temperature. | Vapour Pressures in mm. | ||||
PB Chloro-benzene. | PA Bromobenzene. | PB-760. | Pb-Pa. | M. | |
132° | 760.25 | 395.1 | 0.25 | 365.15 | 0.07 |
134 | 802.15 | 418.6 | 4215 | 383.55 | 10.99 |
136 | 845.85 | 443.2 | 85.85 | 402.65 | 21.32 |
138 | 891.4 | 468.9 | 131.4 | 422.5 | 31.10 |
140 | 938.85 | 495.8 | 178.85 | 443.05 | 40.37 |
142 | 988.2 | 523.9 | 228.2 | 464.3 | 49.15 |
144 | 1039.5 | 553.2 | 279.5 | 486.3 | 57.47 |
146 | 1092.9 | 583.85 | 332.9 | 509.05 | 65.40 |
148 | 1148.4 | 615.75 | 388.4 | 532.65 | 72.92 |
150 | 1206.0 | 649.05 | 446.0 | 556.95 | 80.08 |
152 | 1265.8 | 683.8 | 505.8 | 582.0 | 86.91 |
154 | 1327.9 | 719.95 | 567.9 | 607.95 | 93.41 |
156 | 1392.3 | 757.55 | 632.3 | 634.75 | 99.61 |
In the diagram (Fig. 22) the values of m have been plotted against the temperatures, and the curve is drawn through the points which are not themselves indicated.
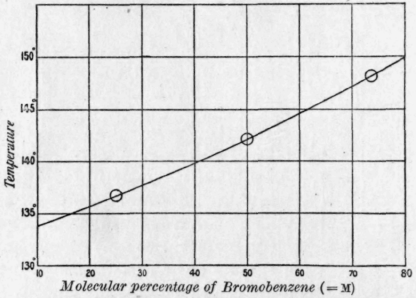
Fig. 22. - Boiling points of mixtures of bromobenzene and chlorobenzene.
Continue to:


