Gas. Part 5
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Gas. Part 5
It has, however, been explained that the exhauster is placed between these two pieces of apparatus. A drawing of the machine has been omitted in the cut from want of room, but the reader can supply the omission. In the latter there is an orifice from which a pipe conducts the gas to the meter, from which it passes through the pipe o to the large reservoir, a small part of which is shown in the figure. In the following table of the constituents of purified coal gas, that in the first two columns is ordinary coal gas from Chemnitz, Saxony; in the third column, ordinary London coal gas; and in the fourth, London cannel coal gas:
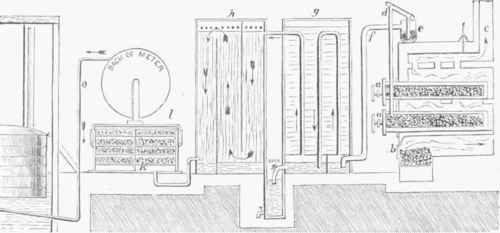
Fig. 7.-Synoptical Plan of Gas Works.
CONSTITUENTS. | 1. | 2. | 3. | 4. |
Hydrogen... | 51.29 | 50.08 | 46.0 | 27.7 |
Marsh gas....... | 86.45 | 85.92 | 89.5 | 50.0 |
Carbonic oxide........ | 4.45 | 5.02 | 7.5 | 6.8 |
Oleflant gas (elavl)..... | 4.91 | 5.33 | 3.8 | 13.0 |
Nitrogen.... | 1.41 | 1.89 | 0.5 | 04 |
Oxygen...... | 0.41 | 0.54 | ........ | .......... |
Carbonic acid.......... | 1.08 | 1.22 | 0.7 | 0.1i |
Aqueous vapor...... | .......... | ----- | 2.0 | 2.0 |
There are other heavy hydrocarbon gases besides elayl, or olefiant gas, as acetylene, trityl, and ditetryl, and also a small quantity of hydrocarbon vapors, which yield light; but the illuminating power of the gas may be regarded as depending principally upon the amount of olefiant gas (heavy carburetted hydrogen) which it contains, the bulk of other gases being carriers rather than light-producers. The olefiant gas is separated by ignition into marsh gas (light carburetted hydrogen) and carbon, the solid particles of which become incandescent and emit white light, which is observed in the luminous cone of a gas flame, and which has the same constitution as that of a candle. (See Flame.) Of the impurities, tar is separated in the hydraulic main and the condenser; ammonia in the hydraulic main, condenser, and washer; sulphuretted hydrogen, cyanogen, and carbonic acid in the washer and lime and iron purifier, a small quantity of the last named gas remaining. The luminosity of a gas flame depends both upon the percentage of heavy hydrocarbons it contains, and the amount of atmospheric air or oxygen mixed with it.
Sometimes in passing it through many purifying processes a small amount of air is absorbed, the oxygen of which, combining with the carbon at the moment of ignition, causes an increased production of heat but diminution of light, on the principle of a Bunsen's burner. The illuminating power may therefore be estimated by analysis; but the practical method is to burn it in comparison with some light-producing body of known power, as a spermaceti candle. This test is made with an instrument called a photometer, a common form of which is shown in fig. 8. An argand burner which consumes a certain number of cubic feet per hour (in experiments usually five feet) is placed at a, and a candle at c. Between them there is a horizontal graduated scale which supports a slide, b, bearing a ground glass screen having a figure in the centre more transparent than the rest of the plate. When this screen is moved to a point on the scale where the figure appears equally bright on each side, the light received from each source will be equal. If two candles are used, placed side by side, and the distance between them and the screen is one eighth that between the latter and the gas-burner, it will show that the light from the burner is equal to that of 16 candles.
A simple screen may be used, or it may have a mirror placed upon each side at the further edge, at such an angle that the two will reflect images of the figure toward the observer, so that a comparison may be made at the same instant. Gas as usually furnished is estimated, when burning at the rate of five cubic feet per hour, to produce a light equal to that given in the same time by 16 or 18 standard sperm candles, each burning at the rate of 120 grains per hour.-The illuminating power of gas depends much upon the form of the burner. It is a matter of common observation that gas may be nearly deprived of its illuminating power if made to issue from the burner with great velocity, or if burned in a tall chimney which produces a very rapid current of air. Very small or thin flames also do not afford conditions of economical expenditure. The smaller or thinner the flame, the greater is its exposure to the oxygen of the air, and consequently the more rapid the consumption of the solid particles of carbon; in other words, the more nearly are the conditions present which cause the flame of a Bunsen's burner to be nearly non-luminous. The conditions to be sought for in an illuminating flame are those which are most conducive to high heating of the carbon particles and to the keeping of them for the longest possible time in an uncombined state, but eventually insuring their complete combustion.
A certain thickness of flame is therefore desirable. A poor gas, if burned in a jet issuing from a wide slit, may be made to yield a better light than a rich gas burned in a very thin flame, which is frequently the fault of the fish-tail burner. According to Prof. Silliman, the illuminating power of a given sample of gas burned in an argand burner is not in the proportion of gas consumed, but more nearly in the proportion of the square of the quantity.-A ton of 2,000 lbs. of good cannel coal, carefully distilled, will yield about 8,000 cubic feet of purified illuminating gas. Other bituminous coals yield from 6,000 cubic feet up to this amount. Every section of a gas works has one or more meters for measuring the volume of gas before it passes into the reservoirs. They are in the form of cylinders, usually about 12 ft. in diameter and from 8 to 12 ft. long. Fig. 9 is a transverse section showing the principle upon which they act. The outer cylinder or drum is stationary. The inner cylinder, turning upon a hollow axis, is divided by the partitions a, a, a, a into five compartments, one in the centre of the hollow axis, and four, d, d, d, d, spiral in direction, exterior to this; slits being left open at e, e, e, e for the passage of the gas from the inner cylinder to the space between it and the outer one, from which it has exit.
Continue to:


