Galvanism, Or Voltaic Electricity. Part 2
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Galvanism, Or Voltaic Electricity. Part 2
After separation the plates will not present the same appearance as during connection; but the evolution of hydrogen gas on the surface of the copper will cease, and if the ends of the wires are examined with a delicate galvanoscope or electroscope, they will be found to contain a small charge of statical electricity, the one connected with the copper plate being in a positive, and the one connected with the zinc plate in a negative condition. If the disconnected ends of the wires are dipped near to each other in a small cup containing a solution of iodide of potassium, the salt will be decomposed; a fact which offers strong evidence against the contact theory. If, in the above experiments, sulphuric instead of hydrochloric acid is used, the same phenomena will appear; hut instead of chloride, sulphate of zinc will be found in solution. A system consisting of two metals immersed in a fluid which chemically acts upon one of them, is called a simple galvanic or voltaic couple, or element. When the plates are disconnected the couple is said to be open, and when connected by a conductor it is said to be closed, forming a circuit, which is also spoken of as open or closed.
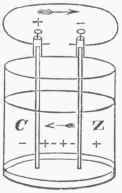
Fig. 1.
When the circuit is closed it is assumed that a current of positive electricity passes through it from the zinc to the copper in the liquid, and from the copper to the zinc out of the liquid. It is assumed that the current passes in this direction because when the end of the wire which is connected with the copper plate is examined with the electroscope, positive electricity is found upon it; and also from the fact that the needle of a galvanometer, when one of the electrodes of the instrument is connected with the copper and the other with the zinc plate, is deflected in the same direction as when they are respectively connected with the prime conductor and rubber plate of an ordinary glass-plate electrical machine. It may be assumed, moreover, that a current of negative electricity passes at the same time in the opposite direction; but for convenience of explanation this assumed action is not taken into consideration. The use of the word current has its advantages, and helps to convey ideas which are in accordance with observed effects; but the actual passage of a fluid in either direction is a matter of doubt, and in the opinion of Faraday does not take place, he believing that the resulting phenomena are caused by a polarization of the molecules of the medium.
The action which is called a voltaic current does not require that one of the metallic elements shall not be at all acted upon by the liquid, but only that the action shall be greater upon one than upon the other; the metal most acted upon being the positive or generating plate. Generally the polarization which results from the connection of two metals when immersed in a dilute acid or saline solution, although either might be acted upon by it separately, causes one to become more and the other less positive, or in other words, more negative, so that it becomes protected. On this principle iron is often protected from corrosive action by coating it with zinc, and the copper bottoms of ships have been in a similar manner protected from the action of sea water by attaching to them a more positive metal. The force which results from a difference in chemical action of a liquid on two metals is called the electromotive force, and its quantity depends not only upon the relative attraction of the metals for constituents of the liquid, but also upon the distance of the plates from each other.
That metal which has the strongest affinity for oxygen is usually the most electro-positive, and one metal may therefore bear an electro-positive relation to a second, while it is electro-negative when compared to a third. Potassium is the most electro-positive of all bodies, but its attraction for oxygen is so violent as to make it practically useless as an element in the galvanic circuit. Among those which can be usefully employed as electro-positive elements, zinc ranks first, while platinum is the most highly electro-negative metal. But the relative electrical condition of several of the metals changes when immersed in different liquids; thus if an iron and a copper plate be connected with the electrodes of a galvanometer and immersed in dilute sulphuric acid, the needle will be deflected in one direction; while if the plates are immersed in a solution of sulphide of potassium, the deflection will be in the opposite direction. The following table shows a few of the results obtained by Faraday:
Comparison Of Different Metals In The Presence Of Different Liquids
Dilute sulph. acid. | Hydrochloric acid. | Sol. of potash. | Sol. sulphide of potash. |
Silver. | Silver. | Iron. | |
Copper. | Silver. | Nickel. | Nickel. |
Antimony. | Nickel. | Copper. | Bismuth. |
Bismuth. | Bismuth. | Iron. | Lead. |
Nickel. | Copper. | Bismuth. | Silver. |
Iron. | Iron. | Lead. | Antimony. |
Lead. | Lead. | Antimony. | Tin. |
Tin. | Tin. | Cadmium. | Copper. |
Cadmium. | Cadmium. | Tin. | Zinc. |
Zinc. | Zinc. | Zinc. | Cadmium. |
The order in each column places the most positive metal in regard to the fluid at the bottom, and the most electro-negative at the top. It has been demonstrated by Poggendorff that the electromotive force between any two metals is equal to the sum of the electromotive forces between all the intervening metals.-The theory of the production of the galvanic current is as follows: When a zinc and a copper plate are immersed in dilute acid, they immediately become polarized, assuming opposite electrical states, that end of the wire, as has been observed, connected with the copper plate showing positive, and that in connection with the zinc negative electricity, while those portions of the copper and zinc plates which are in the liquid are respectively in a negative and a positive state. The compound molecules of the liquid are also supposed to assume a highly polarized condition; one constituent becoming negative and attracted by the zinc, and the other positive and attracted by the copper plate.
Let us suppose the liquid to be hydrochloric acid; the electrical condition and chemical action which take place may be represented as follows:
Continue to:


